- ARAB NEWS
- 02 Jul 2025

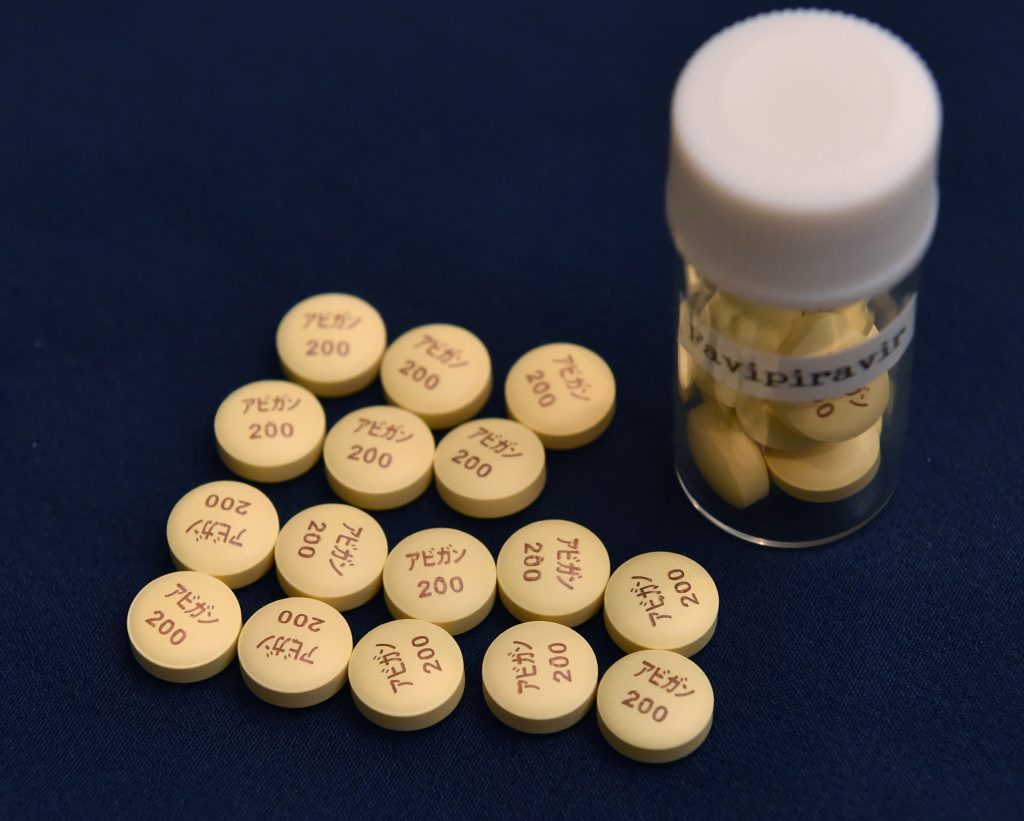
TOKYO: Japanese Prime Minister Shinzo Abe said Monday that his government aims to approve the use of the influenza drug Avigan for treatment of patients with the coronavirus within this month.
Abe made the comments at a press conference held to discuss the government's decision to extend its coronavirus state of emergency through May 31.
Clinical trials are underway to determine whether Avigan can benefit patients with COVID-19, the respiratory disease caused by the coronavirus.
The drug, developed by Fujifilm Toyama Chemical Co., a unit of Fujifilm Holdings Corp., and is expected to benefit coronavirus patients with minor symptoms. But it cannot be used on pregnant women due to concerns about birth defects.
A Japanese medical institution started using Avigan in February to treat people infected with the novel coronavirus, health minister Katsunobu Kato said.
The drug was used for treatment as part of an observation study in February after the consent of the patients was obtained and other related procedures were taken at the medical institution.
Avigan is effective in curbing the growth of the virus and two medical institutions have been preparing to use it, according to Kato.
JIJI Press